Dr. Jai N. Gupta is the founder of EER Systems, Inc., an organization that has been hailed as one of the top 10 defense contractors in the United States. In 2001, L-3 Communications acquired EER Systems, Inc., for $110 million. A skillful entrepreneur and manager, Dr. Jai N. Gupta also served as the chairman and CEO of Centerline Biomedical, a company affiliated with the Cleveland Clinic in northern Ohio.
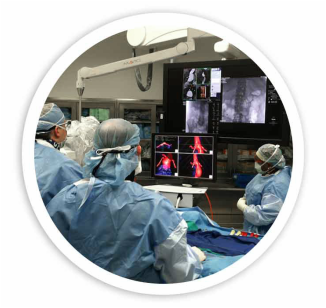
Centerline Biomedical recently developed a cutting-edge device called the Intra-Operative Positioning System (IOPS) that helps surgeons manipulate guidewires and catheters in the body. Traditionally, surgeons have used X-ray fluoroscopy to accomplish these tasks. X-ray fluoroscopy is generally safe, but it presents some risks to patients because of its use of radiation. The amount of radiation that each patient is exposed to varies depending on the type and length of the procedure. IOPS, which is currently in the testing stage, does not emit any radiation. Dr. Matthew Eagleton, a surgeon at Cleveland Clinic who conducted a study with the device, believes that IOPS is a "game-changer" for endovascular therapy.
Centerline hopes that IOPS can enter the market with approval from the Food and Drug Administration (FDA) in late 2017. More information on IOPS is available at goo.gl/9KEEWI
Centerline Biomedical recently developed a cutting-edge device called the Intra-Operative Positioning System (IOPS) that helps surgeons manipulate guidewires and catheters in the body. Traditionally, surgeons have used X-ray fluoroscopy to accomplish these tasks. X-ray fluoroscopy is generally safe, but it presents some risks to patients because of its use of radiation. The amount of radiation that each patient is exposed to varies depending on the type and length of the procedure. IOPS, which is currently in the testing stage, does not emit any radiation. Dr. Matthew Eagleton, a surgeon at Cleveland Clinic who conducted a study with the device, believes that IOPS is a "game-changer" for endovascular therapy.
Centerline hopes that IOPS can enter the market with approval from the Food and Drug Administration (FDA) in late 2017. More information on IOPS is available at goo.gl/9KEEWI
 RSS Feed
RSS Feed
